Highlights:
- Fisetin ameliorates cognitive impairment in rats modeling sepsis – the body’s extreme response to infection, which triggers uncontrolled inflammation and multi-organ dysfunction.
- By promoting the removal of defective mitochondria (mitophagy) and the clearance of compounds that induce oxidative stress, fisetin limits the inflammatory response.
- The reduced inflammatory response in the brain caused by fisetin likely preserves cognition in septic rats.
Although evolution has trained our body’s internal soldiers — white blood cells — to fight off infection effectively with all its got, one particular condition known as sepsis piggybacks off our cellular defense mechanisms to target the whole body. When our bodies are introduced to foreign invaders, cells carefully trigger an inflammatory response. But humans with sepsis, uncontrolled inflammation in response to infection, experience persistent, uncontrolled inflammation, making the body susceptible to unintentional tissue damage and potential multiorgan failure and even death. Notably, nearly half of septic patients succumb to sepsis-associated encephalopathy (SAE), a neurological disorder that occurs secondary to infection in the body without overt central nervous system (CNS) infection.
According to a study published in CNS Neuroscience & Therapeutics by Ding and colleagues, a compound naturally found in plants named fisetin promotes neuroprotective effects in rats with SAE, leading to improved learning and memory formation. The researchers from China find that fisetin activates the removal of defective mitochondria (mitophagy) and eliminates oxidative stress, leading to the inactivation of a multiprotein complex that contributes to the body’s inflammatory response to infection. By blocking this inflammatory multiprotein complex, known as the NLRP3 inflammasome, fisetin blocks the secretion of a specific proinflammatory molecule (IL-1β) into the CNS and protects against cognitive impairments induced by sepsis.
What Do We Already Know About Fisetin?
Fisetin’s medicinal applications are no stranger to the scientific community. Among fisetin’s varying pharmacological properties, one that has gained tremendous attention is fisetin’s ability to target and eliminate non-proliferating (senescent) cells, a key hallmark of aging. Studies also show that the naturally abundant flavonol is an effective antioxidant and anti-inflammatory agent, with documented chemotherapeutic applications. More importantly, researchers have successfully utilized fisetin as a neuroprotective agent to halt the progression of harmful neurological diseases, including Alzheimer’s and Parkinson’s disease. While there’s excitement in knowing fisetin’s health benefits, scientists are eager to identify the underlying mechanisms that govern these events.
Fisetin Rescues Cognitive Dysfunction in Rats with SAE
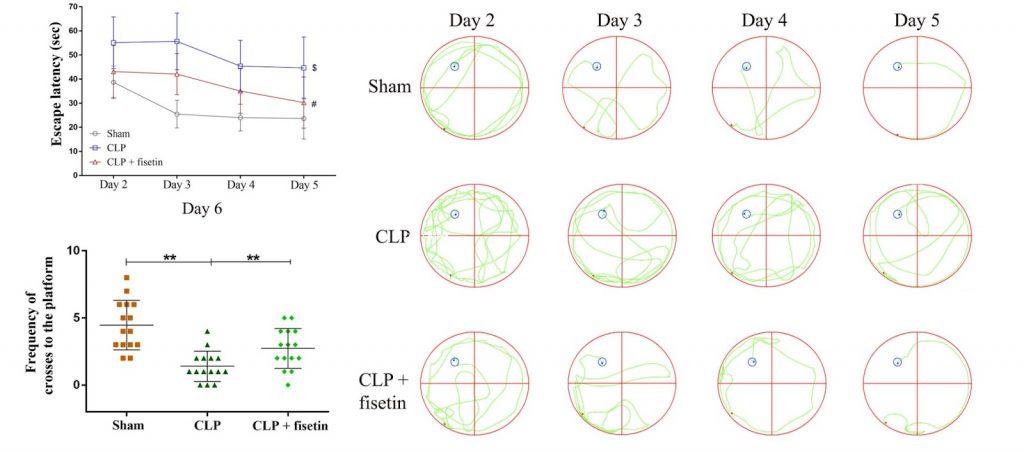
To evaluate the effects of fisetin on learning and memory formation, Ding and colleagues performed the Morris Water Maze Test. Here, rats are trained to locate a hidden platform submerged in a pool of water divided into four quadrants lined with spatial cues. By looking at the time it takes for rats to find the hidden platform (escape latency), the investigators can get a readout of the quality of spatial memory in these rodents.
After four days of observations, Ding and colleagues saw that the escape latency was much higher in septic rats than control rats, indicating that SAE altered cognition. However, supplementing fisetin (20 mg/kg) to septic rats significantly decreased escape latency over the four days, suggesting that fisetin attenuated SAE-induced memory deficits. What’s more, the escape passage — defined by the distance traveled to locate the hidden platform, substantially decreased after supplementing fisetin to septic rats — further indicating that fisetin contributes to improved learning capabilities and memory formation in SAE rats.
Fisetin Ameliorates Neuroinflammation by Clearing Mitochondrial Damage and Oxidative Stress
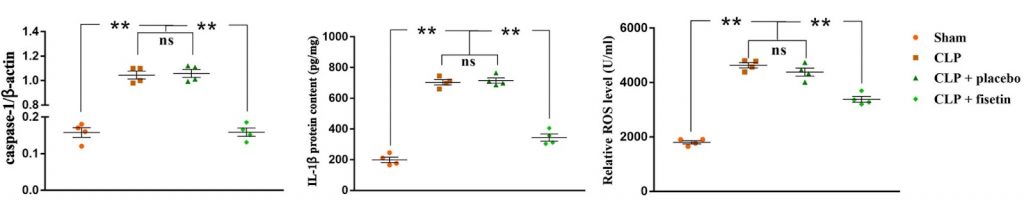
Next, Ding and colleagues determined the root cause of neuroinflammation, which is associated with cerebral dysfunction in septic patients. They proposed that sepsis promoted NLRP3 inflammasome activation, resulting in the secretion of a proinflammatory molecule (IL-1β) into the CNS. Notably, oxidative stress, which can arise from defective mitochondria, promotes NLRP3 activation. The research team from the Guangdong Academy of Medical Sciences found that septic rats exhibited significantly higher ROS levels compared to control rats. What’s more, they located impaired mitochondria in septic rats, highlighting the possibility that sepsis mediated mitochondrial damage, leading to increased ROS production.
Upon confirming that sepsis increased oxidative stress, Ding and colleagues proceeded with analyzing IL-1β protein expression levels in septic rats to see if ROS activated the NLRP3 inflammasome. As expected, IL-1β protein content was much higher in septic rats compared to control rats, confirming that sepsis-induced oxidative stress triggers NLRP3 activation and promotes IL-1β secretion into the CNS, resulting in neuroinflammation and brain dysfunction.
Together, these findings link fisetin’s effect on rescuing spatial memory to improved neuroinflammation in septic rats, as evidenced by the significant decrease in IL-1β protein levels. Notably, investigators failed to locate any mitochondrial damage after fisetin administration. However, what they did find were autophagosomes, compounds that assist with the removal of damaged mitochondria, indicating that fisetin promotes mitophagy. Given that ROS levels also plummeted following fisetin administration, investigators proposed that ROS elimination likely resulted from mitophagy activation. Taken together, the investigators suggest that fisetin ameliorates neuroinflammation and rescues cognition in septic rats by eliminating oxidative stress via mitophagy, which blocks NLRP3 activation and prevents the secretion of IL-1β into the CNS.
Are There Clinical Trials Evaluating the Effectiveness of Fisetin on Brain Inflammation?
Not only are scientists continually evaluating fisetin in rodents, but several human clinical trials are ongoing that are still in their infancy. Although there aren’t any active clinical trials evaluating the protective nature of fisetin in sepsis patients, the senolytic is being tested for its effectiveness in ameliorating several conditions that induce frailty and inflammation in elderly adults. Given the results of this study and the fact that there are other senolytics being actively tested for their role in human brain health, there is ample evidence for testing fisetin in this context.