Key Points:
- CML, a metabolite, increases within microglia with age, leading to increased cellular stress, decreased mitochondrial function, and decreased cellular energy.
- The gut bacteria (the intestinal microbiome) profile changes with age leading to increased intestinal permeability.
- These intestinal permeability changes drive the accumulation of CML within the brain and bloodstream.
Numerous studies of the gut-brain axis suggest that our bacterial profiles can affect anything from mood to immunity. Now researchers are turning to microglia – which function as macrophages, removing damaged cells and infections from the central nervous system (the spine and brain) – to determine how our microbiome may affect their functionality. Microglial age-related changes that lead to decreased functionality are mediated through interactions between the central nervous system and the external environment, including bacteria within the gut.
A study out of Germany, published in Nature Neuroscience, provides evidence that as the intestinal microbiome changes with age, intestinal permeability increases, allowing specific metabolites – or byproducts of nutrient breakdown – to enter the bloodstream and make their way to the brain negatively affecting microglial function. They highlight a specific metabolite, N6-carboxymethyllysine (CML), which may be responsible for many of the age-related microglial changes, including the decreased function of mitochondria (the cell’s powerhouse), decreased cellular energy, and increased cellular stress via reactive oxygen species.
“Ensuring intestinal barrier integrity to regulate the translocation of metabolites from the gut may represent a new approach to promoting brain health,” John W. Bostick and Sarkis K. Mazmanian said in an editorial about the research.
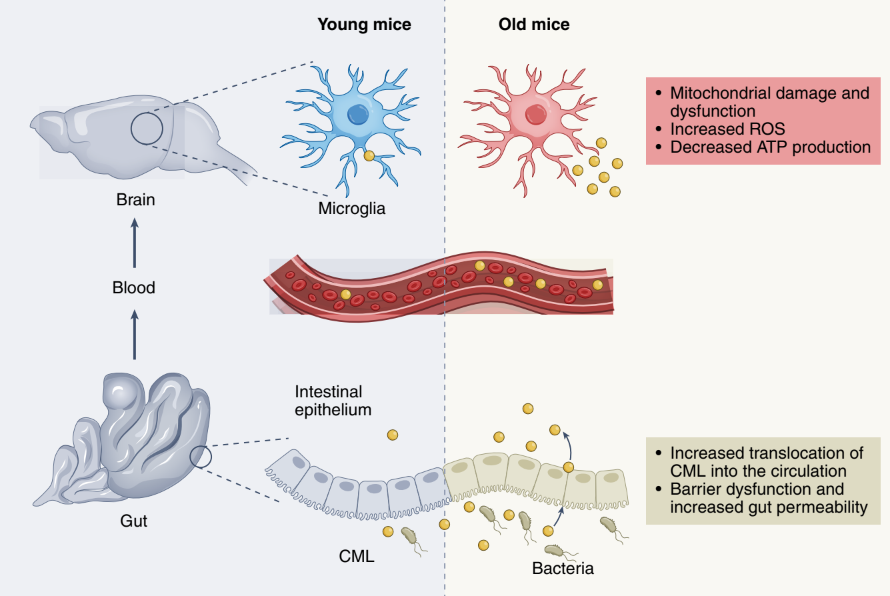
Metabolites from the Nutrient Breakdown in the Gut Lead to Decreased Microglial Function Via Increased Intestinal Permeability
For their studies, the researchers used two kinds of mice: one with a screened, healthy profile of gut bacteria called SPF and another completely lacking gut bacteria called GF. The investigators found increased reactive oxygen species in aged SPF mice. These reactive oxygen species, which are a major driver of cellular aging through increased cellular stress, led to increased damage to and dysfunction of the microglial mitochondria. This resulted in decreased mitochondrial activity and cellular energy as the mice aged, indicating the role that intestinal bacteria may play in regulating cellular stress in the brain.
The Caltech scientists found that CML was elevated in the bloodstream and brains of the aged SPF mice, which led to increased age-related microglial dysfunction. In contrast, the natural levels of CML in the brains of aged GF mice were similar to those of young adult mice, suggesting that gut bacteria are needed for increased CML levels in the aged brain. Additionally, when young mice were given CML, the treatment increased cellular stress, reduced cellular energy stores, and caused mitochondrial dysfunction, partially recapitulating the changes in microglia of aged mice.
Further investigation revealed that age-related microbiome changes increased intestinal permeability, leading to more CML leaking into the bloodstream, allowing it to get to the brain and induce microglial dysfunction. This did not occur in the GF mice, and young adult mice were only minorly affected because most of their CML stays in their intestines and is excreted due to their intact intestinal wall. In fact, the investigators found that when the gut barrier function was enhanced through medication, the aged SPF mice had less gut leakiness, less CML accumulation in the brain, reduced microglial reactive oxygen species, and increased energy within the microglial cells.
How our Environment Impacts our Aging
This research highlights how much our gut microbiome impacts the aging of our bodies. More research is needed to determine the impact of these findings. Still, if our gut microbiome is so crucial to aging and its changes can mediate impacts on brain function, maybe ways of restoring our gut microbiome to its youth or maintaining its barrier are the key to holding onto our brain function as we age.
“These findings underline the impact of age-induced microbiota alterations, which disrupt the integrity of the gut barrier and facilitate the accumulation of CML in the brains of aged mice and humans,” the researchers wrote.
The use of probiotics and fecal transplants for those suffering from intestinal distress is not unheard of, but maybe research like this will bring these treatments into the realm of anti-aging protocols. Possibly in the not too distant future, we’ll be able to go to the spa and get botox injections, as well as fecal matter transplants from younger, healthy individuals, or maybe we’ll just stick to medications that enhance our intestinal barrier function.