Aging takes place in many animals, and humans are no exception. But what does biological aging actually mean?
Researchers describe aging as a reduction in the functional capabilities occurring over time and that typically begin once we become sexually mature. Changes affecting the function and activity of cells called biological hallmarks of aging cause deteriorating performance of everyday tasks, like taking out the trash or remembering where you left your car keys. Although we’re getting a better handle on the effects that aging has on cells, the variations in the inherited DNA sequence that contribute to these biological hallmarks of aging has not been fully uncovered.
In a review published in Nature Reviews Genetics, Melzer and colleagues from the University of Connecticut and the National Institute on Aging looked at how scientists use genetics research to uncover what happens inside cells during human aging. This research can help us understand and figure out how to sustain healthier, longer lives and reduce periods where people experience disability.
Linking DNA Variants to Aging
In their review, Melzer and colleagues present findings from genome-wide association studies (GWAS), a method used in genetics research to link DNA sequence variations from many people to particular diseases. In examining the DNA variations associated with longevity, these studies zeroed in on a specific gene that plays a role in longevity called APOE. Interestingly, this gene codes for a protein involved in transporting cholesterol to cells and functions in the brain to maintain and repair neuron connections, which are called synapses. So, a link likely exists between certain inherited DNA sequences in this gene and the onset and impact of Alzheimer’s and heart disease.
Melzer and colleagues also presented analyses of DNA variants playing significant roles in influencing longevity and aging as well as multiple age-related diseases like Alzheimer’s or coronary artery disease. They uncovered 22 genetic variants linked with multiple age-related diseases, 12 of which are mostly inherited together. One of these variants was found in APOE and was linked with Alzheimer’s and coronary artery disease. Two other DNA variants were located in the genes LPA and LDLR and were linked to altered blood fat levels and cardiovascular traits. The remaining nine associations between genetic variants that tend to get inherited together and age-associated diseases included links to three or more of the diseases, including Alzheimer’s disease, stroke, coronary artery disease, type 2 diabetes, kidney disease, osteoarthritis, and several cancers.
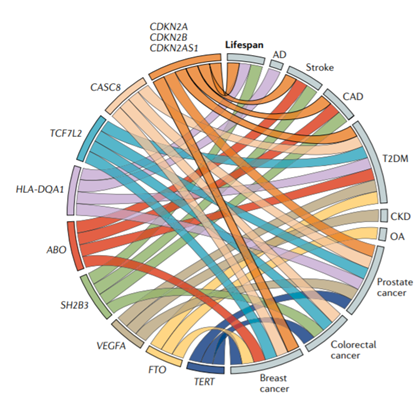
(Melzer et al., 2020 | Nature Reviews. Genetics) Genetic variants with tendencies to get inherited together in nine genes were linked with multiple age-related diseases. The associations of DNA variants from these genes with specific diseases are illustrated in the diagram as colored lines. AD, Alzheimer’s disease; CAD, coronary artery disease; CKD, chronic kidney disease; OA, osteoarthritis; T2DM, type 2 diabetes mellitus.
Determining how human aging occurs at the DNA level and in cells remains in its infancy. The ability to balance DNA damage with repair processes is emerging as a driving force behind aging. Data from GWAS studies indicate that genetic variants linked with age-related diseases play influential roles in these processes.
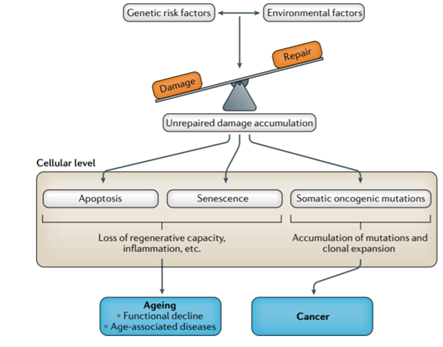
(Melzer et al., 2020 | Nature Reviews. Genetics) Mechanisms of human aging. Aging processes come from a balance of physiological damage and repair mechanisms. The impact of DNA damage grows with chronological age as DNA repair capabilities diminish, causing cells to die (apoptosis), stop replicating (senescence), or accumulate cancer-causing mutations. All of this results in an elevated onset of chronic diseases from aging or cancer.
“Human genetics will likely continue to provide growing insights into how we age and play a major role in identifying ways in which we might slow ageing, thus helping more people to age well,” stated Melzer and colleagues in their review. Researchers have begun to define which genes lead to aging which may lead to therapeutic options soon. Understanding the roles genetics plays can lead to treatments facilitating longer lives while diminishing periods of disability and disease.