Although the immune system utilizes inflammation as a method to restore health in the body, there is evidence suggesting that inflammation may be linked to progressive heart failure. Immune cells that facilitate this process highly rely on the mitochondria, the powerhouse of the cell, to provide energy. However, researchers still don’t know whether malfunctional mitochondria in various circulating immune cells amplify inflammation in patients that exhibit heart failure.
Clinical trial (NCT03727646) results were published by Zhou and colleagues from the University of Washington in the Journal of Clinical Investigation, and the presented data elucidated the relationship between heart failure, inflammation, and mitochondria in immune cells. The research team identified a connection between heart inflammation in patients and malfunctional mitochondria in peripheral blood mononuclear cells (PBMCs), a type of immune cell. When patients that exhibited heart failure underwent oral nicotinamide riboside (NR) supplementation (1000 mg twice daily), PBMC mitochondrial function was improved, and inflammation decreased. NR is a precursor to nicotinamide adenine dinucleotide (NAD+), an essential energy-producing compound. The obtained data from the study propose that preventative measures can be taken to combat heart disease through NAD+ augmentation.
NAD+ Helps Hearts Heal in Mice
Mice have been shown to exhibit decreased inflammation and greater protection of the heart upon increased levels of NAD+, which corresponds to optimal mitochondrial function. These current findings suggest the possibility of improving the health of patients with heart failure by boosting levels of NAD+, which allows for inflammation to be modulated. However, researchers still do not know if circulating immune cells can reduce inflammation in heart patients by improving mitochondrial function by elevating NAD+ levels.
Does NAD+ Protect Human Patients with Heart Disease?
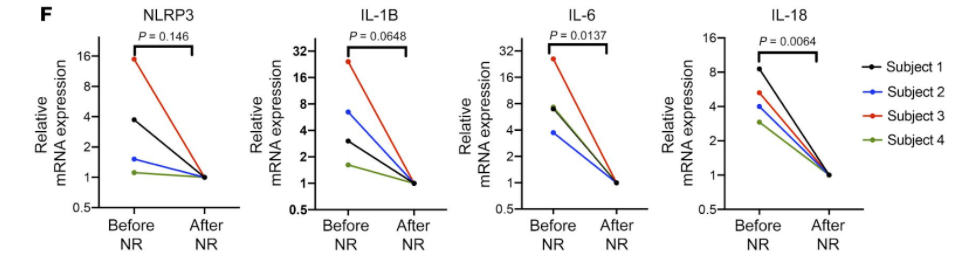
Zhou and colleagues differentiated mitochondrial function of PBMCs in 19 healthy patients and mitochondrial function of PBMCs in 19 hospitalized patients that exhibited heart failure. Diminished mitochondrial function was observed in PBMCs of heart failure patients. These cells also showed higher concentrations of proinflammatory cytokines, small enzymes that are pivotal in modulating the growth and activity of cells in the blood and immune system.
Human heart failure patients were orally supplemented with nicotinamide riboside (NR), an NAD+ precursor, for five to nine days. Zhou and colleagues obtained blood samples from these patients before and after the trial and examined each blood sample’s contents. Furthermore, the following escalating NR dosages were used to treat five hospitalized heart failure patients for five to nine days: day 1- 250 mg twice a day, day 2- 500 mg twice a day, and day 3 to day 9- 1,000 mg twice a day.
Results showed that patients who exhibited heart failure demonstrated improved mitochondrial function after utilizing NR to raise NAD+ levels. The production of proinflammatory cytokines was also decreased in heart failure patients. Investigators suggest that NR supplementation induced the preservation of mitochondrial function and diminished the production of cell harming compounds called reactive oxygen species in the presence of a compound that modulates inflammation.
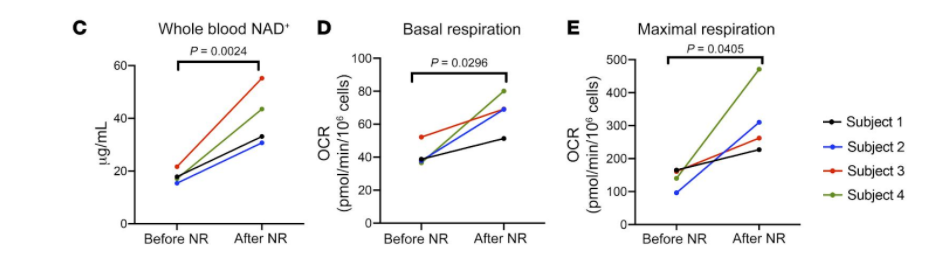
Zhou and colleagues illustrate how the activation of peripheral immune cells in human heart failure patients through inflammation heavily depends on the function of mitochondria. The study presented further suggests that mitochondrial function can be ameliorated by elevating NAD+ levels, which also leads to reduced inflammation caused by PBMCs in patients that exhibit heart failure. In addition, the inactivation of circulating immune cells through blockage of proinflammatory cytokines allows for inflammation to be halted and leads to the collapse of the harmful cycle perpetuating the disease.
More Clinical Trials on NAD+ and Heart Failure are Needed
However, there are several limitations to this study. Both the small size of experimental groups and the short treatment periods of the experiments (five to nine days) involving supplementation of NR to heart failure patients limit the investigators in formulating plausible conclusions concerning the safety and efficacy of NR. The findings of this study show promise in evaluating NR’s potential anti-inflammatory effect in future clinical trials.
“To our knowledge, this study is the first to examine whether NAD+ augmentation has anti-inflammatory effects in the context of human heart failure,” said the investigators in their article. “Although the human efficacy data are currently lacking, the collective actions of NAD+ repletion on preserving cardiac function and reducing systemic inflammation may provide a molecular basis for a novel heart failure therapy.”