Highlights
- Using CRISPR to screen critical genes in the cancer-eliminating immune response, researchers from ShanghaiTech University identify a role for NAD+ metabolism.
- Increasing NAD+ levels by supplementing with nicotinamide improves anti-cancer CAR-T immunotherapy in mice, extending their lifespans.
- This study establishes a conceptual basis for enhancing tumor immunotherapy through simple nutritional supplements that boost NAD+ levels.
Cancer has claimed countless lives throughout human history. In developing more effective ways to treat the devastating invasion of tumor cells into surrounding tissues, scientists are eyeing methods to activate immune cells called T cells that target tumors potently. T cells often sit dormant next to growing tumor cells even though they can effectively eliminate the cancerous cells. Identifying how and why T cells often lay in a suppressed state near tumor cells could improve cancer immunotherapies dependent on T cell activation.
Fan and colleagues from ShanghaiTech University in China published a study in Cell Reports showing impaired nicotinamide adenine dinucleotide (NAD+) synthesis in dormant T cells near tumors. Increasing NAD+ levels with an over-the-counter nutrient precursor nicotinamide (NAM) boosted T cell activity and strengthened anti-cancer immunotherapies. These findings offer hope that targeting the NAD+ metabolic pathway may enhance the effectiveness of cancer immunotherapies relying on T cell activation.
NAD+ Synthesis Is Essential for Anti-Tumor T Cell Activation
Since immune cells and tumor cells share numerous metabolic pathways, they compete for nutrients. And the uncontrolled proliferation of tumors can deplete nutrients, potentially including NAD+, and lead to the toxic accumulation of metabolites. This process exhausts T cells and stresses their power-generating cell structures (mitochondria), hindering immunotherapies that target cancer. Because of this push and pull for nutrients, Fan and colleagues proposed that boosting T cell metabolism might restore their inhibited function and reinvigorate their anti-tumor activity.
To test their proposal, Fan and colleagues identified the major metabolic players that fuel the anti-tumor metabolic response in T cells. To do so, the ShanghaiTech researchers performed a genetic screen using CRISPR technology that disrupts genes in T cells inside a laboratory dish and examined T cell activation. This analysis found that the NAMPT gene, which encodes an enzyme essential for NAD+ synthesis, is crucial for proper T cell activation. Their findings suggest that faulty NAD+ metabolism from malfunctioning NAMPT activity might inhibit T cell activation near cancer cells.
NAD+ Supplementation Enhances CAR-T Anti-Tumor Immunotherapy, Improving Mouse Survival
Since dysfunctional NAD+ synthesis may suppress T cell activation, Fan and colleagues tested if enhancing NAD+ levels could improve CAR-T therapy in tumor-ridden mice. CAR-T therapy is the genetic engineering of T cell receptors that identify and bind to tumor cells, giving activated T cells a leg up at eliminating cancer cells. Injecting the mice with the NAD+ precursor NAM significantly elevated NAD+ levels in CAR-T immune cells. Moreover, NAM injections in concert with CAR-T cell therapy stimulated a strong melanoma anti-tumor effect. In fact, the mice were all tumor-free by the end of the 49-day experiment, substantially extending their lifespans.
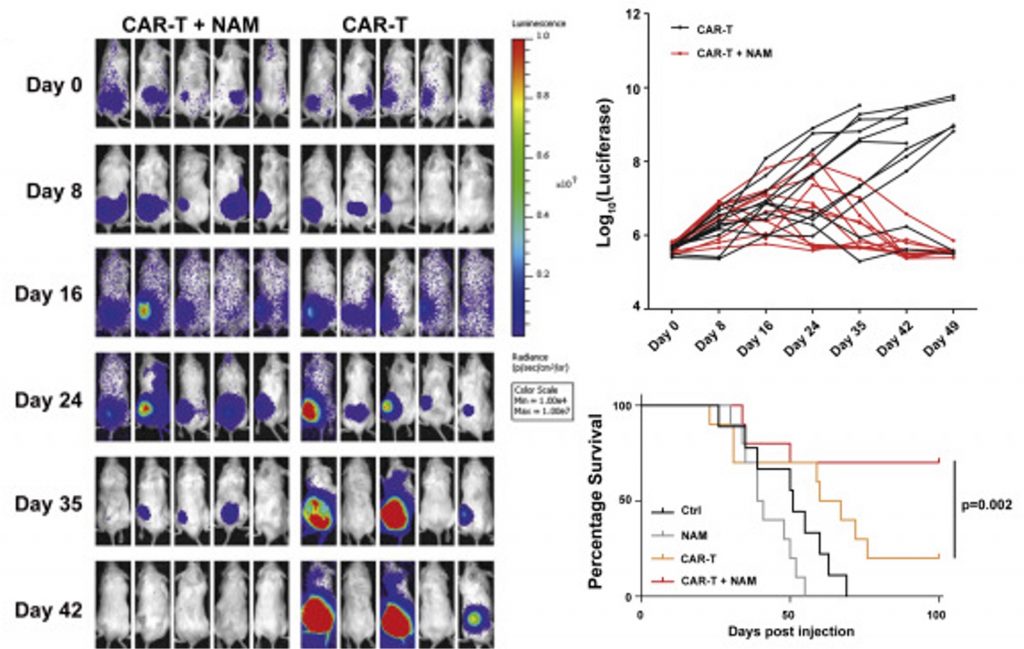
“Taken together, these results establish that supplementation with the NAD+ precursor NAM enhances CAR-T cell tumor killing function and improves the therapeutic efficacy of immunotherapy,” said Fan and colleagues. “We strongly believe that boosting NAD+ concentration in T cells through a variety of genetic and/or pharmacological approaches will better activate T cells, thereby enhancing the efficacy of tumor immunotherapy in the foreseeable future.”
Targeting NAD+ Metabolism to Improve Immunotherapy
NAM supplementation is one way to boost NAD+, but there are other NAD+ precursors available that may be just as potent. For example, a previous report showed that supplementing with the precursor NMN promotes the elimination of immunotherapy-resistant tumors when combined with antitumor PD-L1 antibodies. Moreover, gene therapy that increases NAMPT activity may constitute another means to rescue reduced NAD+ levels in T cells around tumors. As NAD+ level-restoring technologies continue evolving, one thing is clear — NAD+ metabolism is a promising therapeutic target for enhancing the efficacy of anti-tumor immunotherapies.